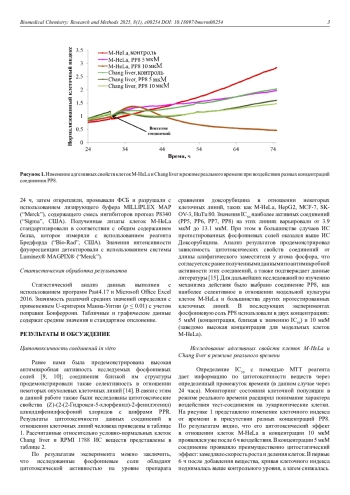
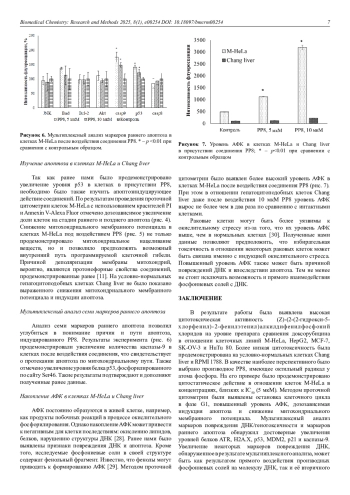
1. Makimbetov, E.K., Salikhar, R.I., Tumanbaev, A.M., Toktanalieva, A.N., Kerimov, A.D. (2020) Cancer epidemiology in the world. Modern problems of science and education, 2, 168-168. DOI: 10.17513/spno.29718
2. Global cancer burden growing, amidst mounting need for services. Retrieved September 30, 2024, from: https://www.who.int/news/item/01-02-2024-globalcancer- burden-growing--amidst-mounting-need-for-services
3. Kaprin, A.D., Starinsky, V.V., Shakhzadova, A.O. (2022). Malignant neoplasms in Russia in 2021 (incidence and mortality). Moscow: PA Hertsen Moscow Oncology Research Institute–Branch of the National Medical Research Radiological Center. 252 p. ISBN 978-5-85502-280-3
4. Peyraga, G., Ducassou, A., Arnaud, F.X., Lizée, T., Pouédras, J., Moyal, É. (2021). Radiothérapie et toxicité médullaire: actualités et perspectives. Cancer/ Radiothérapie, 25(1), 55-61. DOI: 10.1016/j.canrad.2020.05.017
5. Zhu, S., Wang, X., Jiang, H. (2024). Systematic Reversal of Drug Resistance in Cancer. Targets, 2(3), 250-286. DOI: 10.3390/targets2030015
6. Pawar, A., Korake, S., Pawar, A., Kamble, R. (2023). Delocalized lipophilic cation triphenyl phosphonium: promising molecule for mitochondria targeting. Current Drug Delivery, 20(9), 1217-1223. DOI: 10.2174/1567201819666220525092527
7. Ibrahim, M.K., Haria, A., Mehta, N.V., Degani, M.S. (2023). Antimicrobial potential of quaternary phosphonium salt compounds: A review. Future Medicinal Chemistry, 15(22), 2113-2141. DOI: 10.4155/fmc-2023-0188
8. Nissim, M., Lline-Vul, T., Shoshani, S., Jacobi, G., Malka, E., Dombrovsky, A., Banin, E., Margel, S. (2023). Synthesis and Characterization of Durable Antibiofilm and Antiviral Silane-Phosphonium Thin Coatings for Medical and Agricultural Applications. ACS omega, 8(42), 39354-39365. DOI: 10.1021/acsomega.3c04908
9. Tatarinov, D.A., Kuznetsov, D.M., Voloshina, A.D., Lyubina, A.P., Strobykina, A.S., Mukhitova, F.K., Polyancev, F.M., Mironov, V.F. (2016). Synthesis of 2-(2-hydroxyaryl) alkenylphosphonium salts from phosphine oxides via ringclosing ring-opening approach and their antimicrobial evaluation. Tetrahedron, 72(51), 8493-8501. DOI: 10.1016/j.tet.2016.11.023
10. Terekhova, N.V., Lyubina, A.P., Voloshina, A.D., Sapunova, A.S., Khayarov, K.R., Islamov, D.R., Usachev, K.S., Evtugyn, V.G., Tatarinov, D.A., Mironov, V.F. (2022). Synthesis, biological evaluation and structure-activity relationship of 2-(2-hydroxyaryl) alkenylphosphonium salts with potency as anti-MRSA agents. Bioorganic Chemistry, 127, 106030. DOI: 10.1016/j.bioorg.2022.106030
11. Rokitskaya, T.I., Terekhova, N.V., Khailova, L.S., Kotova, E.A., Plotnikov, E.Y., Zorov, D.B., Tatarinov, D.A., Antonenko, Y.N. (2019). Zwitterionic protonophore derived from 2-(2-hydroxyaryl)alkenylphosphonium as an uncoupler of oxidative phosphorylation. Bioconjugate Chemistry, 30(9), 2435- 2443. DOI: 10.1021/acs.bioconjchem.9b00516
12. IC50 Calculator. Retrieved June 11, 2024, from: https://www.aatbio.com/ tools/ic50-calculator
13. Kho, D., MacDonald, C., Johnson, R., Unsworth, C.P., O’Carroll, S.J., Du Mez, E., Angel, C.E., Graham, E.S. (2015). Application of xCELLigence RTCA biosensor technology for revealing the profile and window of drug responsiveness in real time. Biosensors, 5(2), 199-222. DOI: 10.3390/bios5020199
14. Terekhova, N.V., Tatarinov, D.A., Shaihutdinova, Z.M., Pashirova, T.N., Lyubina, A.P., Voloshina, A.D., Sapunova, A.S., Zakharova, L.Ya., Mironov, V.F. (2020). Design and synthesis of amphiphilic 2 hydroxybenzylphosphonium salts with antimicrobial and antitumor dual action. Bioorganic & Medicinal Chemistry Letters, 30(13), 127234. DOI: 10.1016/j.bmcl.2020.127234
15. Chen, Z., Luo, R., Xu, T., Wang, L., Deng, S., Wu, J., Wang, H., Lin, Y., Bu, M. (2024). Design, synthesis and antitumor effects of lupeol quaternary phosphonium salt derivatives. Bioorganic & Medicinal Chemistry, 113, 117934. DOI: 10.1016/j.bmc.2024.117934
16. Abassi, Y.A., Xi, B., Zhang, W., Ye, P., Kirstein, S.L., Gaylord, M.R., Feinstein, S.C., Wang, X., Xu, X. (2009). Kinetic cell-based morphological screening: prediction of mechanism of compound action and off-target effects.Chemistry & biology, 16(7), 712-723. DOI: 10.1016/j.chembiol.2009.05.011
17. Xie, W., Ye, Y., Shen, A., Zhou, L., Lou, Z., Wang, X., & Hu, J. (2008). Evaluation of DNA-targeted anti-cancer drugs by Raman spectroscopy. Vibrational Spectroscopy, 47(2), 119–123. DOI: 10.1016/j.vibspec.2008.03.003
18. Bohgaki, T., Bohgaki, M., Hakem, R. (2010). DNA double-strand break signaling and human disorders. Genome integrity, 1, 1-14. DOI: 10.1186/2041-9414-1-15
19. Ramazanov, B.R., Khusnutdinov, R.R., Galembikova, A.R., Dunaev, P.D., Boichuk S.V. Role of p53 protein in activation of ATM- and PARP-mediated DNA damage repair (DDR) pathways induced by topoisomerase type II inhibitors. Kazan medical journal, 97(2), 245-249. DOI: 10.17750/KMJ2016-245
20. Smith, J., Tho, L.M., Xu, N., Gillespie, D.A. (2010). The ATM–Chk2 and ATR–Chk1 pathways in DNA damage signaling and cancer. Advances in cancer research, 108, 73-112. DOI: 10.1016/B978-0-12-380888-2.00003-0
21. Abbas, I., Badran, G., Verdin, A., Ledoux, F., Roumie, M., Guidice, J.M.L., Courcot, D., Garçon, G. (2019). In vitro evaluation of organic extractable matter from ambient PM2. 5 using human bronchial epithelial BEAS-2B cells: Cytotoxicity, oxidative stress, pro-inflammatory response, genotoxicity, and cell cycle deregulation. Environmental research, 171, 510-522. DOI: 10.1016/j.envres.2019.01.052
22. Ismail, I.H., Hendzel, M.J. (2008). The γ‐H2A. X: Is it just a surrogate marker of double‐strand breaks or much more?. Environmental and molecular mutagenesis, 49(1), 73-82. DOI: 10.1002/em.20358
23. Craig, A., Scott, M., Burch, L., Smith, G., Ball, K., Hupp, T. (2003). Allosteric effects mediate CHK2 phosphorylation of the p53 transactivation domain. EMBO reports, 4(8), 787-792. DOI: 10.1038/sj.embor.embor901
24. Loughery, J., Cox, M., Smith, L.M., Meek, D.W. (2014). Critical role for p53-serine 15 phosphorylation in stimulating transactivation at p53-responsive promoters. Nucleic acids research, 42(12), 7666-7680. DOI: 10.1093/nar/gku501
25. Liebl, M.C., Hofmann, T.G. (2019). Cell fate regulation upon DNA damage: p53 serine 46 kinases pave the cell death road. Bioessays, 41(12), 1900127. DOI: 10.1002/bies.201900127
26. Karimian, A., Ahmadi, Y., Yousefi, B. (2016). Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage. DNA repair, 42, 63-71. DOI: 10.1016/j.dnarep.2016.04.008
27. Liebl, M.C., Hofmann, T.G. (2019). Cell fate regulation upon DNA damage: p53 serine 46 kinases pave the cell death road. Bioessays, 41(12), 1900127. DOI: 10.1002/bies.201900127
28. Reczek, C.R., Chandel, N.S. (2017). The two faces of reactive oxygen species in cancer. Annual review of cancer biology, 1(1), 79-98. DOI: 10.1146/annurev-cancerbio-041916-065808
29. Sui, X., Wang, J., Zhao, Z., Liu, B., Liu, M., Liu, M., Shi, C., Feng X., Fu Y., Shi D., Li S., Qi Q., Xian Mo., Zhao, G. (2024). Phenolic compounds induce ferroptosis-like death by promoting hydroxyl radical generation in the Fenton reaction. Communications Biology, 7(1), 199. DOI: 10.1038/s42003-024-05903-5
30. Wang, Q.Y., Xu, Y.S., Zhang, N.X., Dong, Z.P., Zhao, B.N., Liu, L.C., Lu T., Wang, Y. (2020). Phenylboronic ester-modified anionic micelles for ROS-stimuli response in HeLa cell. Drug Delivery, 27(1), 681-690. DOI: 10.1080/10717544.2020.1748761