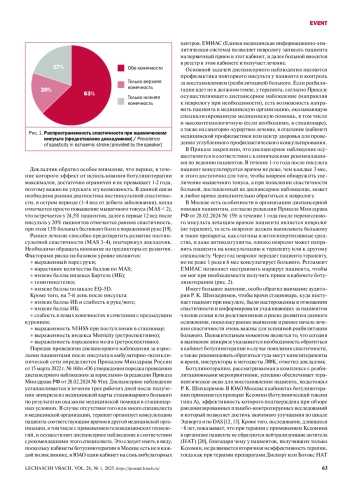
1. Opheim A., Danielsson A., Murphy M. A., et al. Upper-limb spasticity during the first year after stroke: stroke arm longitudinal study at the University of Gothenburg. Am J Phys Med Rehabil. 2014; 93 (10): 884-896. DOI: 10.1097/PHM.0000000000000157
2. Wissel J., Manack A., Brainin M. Toward an epidemiology of poststroke spasticity. Neurology. 2013; 80 (3 Suppl 2): S13-19. DOI: 10.1212/WNL.0b013e3182762448
3. Lundström E., Smits A., Terént A., Borg J. Time-course and determinants of spasticity during the first six months following first-ever stroke. J Rehabil Med. 2010; 42: 296-301.
4. Li Sh., Francisco G. E., Rymer W. Z. A new defenition of poststroke spasticity and the interference of spasticity with motor recovery from acute to chronic stages. Neurorehabil Neural Repair. 2021; 35 (7): 601-610. DOI: 10.1177/15459683211011214 EDN: QFLOWI
5. Marsden J., Stevenson V., Jarrett L. Treatment of spasticity. Handb Clin Neurol. 2023;196: 497-521. DOI: 10.1016/B978-0-323-98817-9.00010-7
6. Triccas L. T., Kennedy N., Smith T., Pomeroy V. Predictors of upper limb spasticity after stroke? A systematic review and meta-analysis. Physiotherapy. 2019; 105 (2): 163-173. DOI: 10.1016/j.physio.2019.01004
7. Gohritz A., Jan Fridén J. Management of Spinal Cord Injury-Induced Upper Extremity Spasticity. Hand Clin. 2018; 34 (4): 555-565. DOI: 10.1016/j.hcl.2018.07.001
8. Halabchi F., Alizadeh Z., Sahraian M. A., Abolhasani M. Exercise prescription for patients with multiple sclerosis; potential benefits and practical recommendations. BMC Neurol. 2017; 17 (1): 185. DOI: 10.1186/s12883-017-0960-9 EDN: KFHTZA
9. Kong K. H., Lee J., Chua K. S. Occurrence and temporal evolution of upper limb spasticity in stroke patients admitted to a rehabilitation unit. Arch Phys Med Rehabil. 2012; 93 (1): 143-148. DOI: 10.1016/j.apmr.2011.06.027
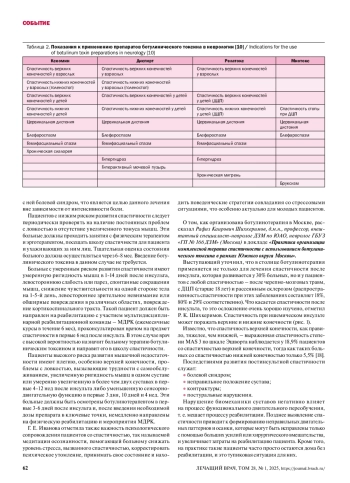
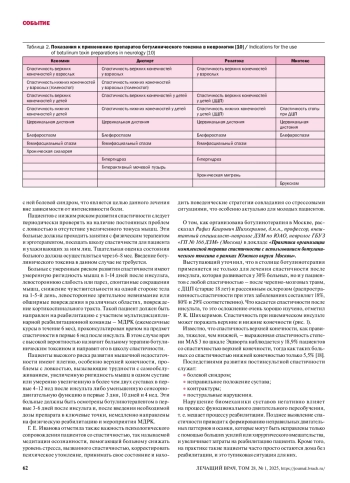
10. Инструкции по применению препаратов, 2024.
11. Kaňovský P., Slawek J., Denes Z., et al. Efficacy and safety of treatment with incobotulinum toxin A (botulinum neurotoxin type A free from complexing proteins; NT 201) in post-stroke upper limb spasticity. J Rehabil Med. 2011; 43 (6): 486-492. DOI: 10.2340/16501977-0796 EDN: PLPOBN
12. Elovic E. P., Munin M. C., Kaňovský P., et al. Randomized, placebo-controlled trial of incobotulinumtoxina for upper-limb post-stroke spasticity. Muscle & Nerve. 2016; 53 (3): 415-421. DOI: 10.1002/mus.24776
13. Kaňovský P., Elovic E. P., Angelika Hanschmann A., et al. Duration of Treatment Effect Using IncobotulinumtoxinA for Upper-limb Spasticity: A Post-hoc Analysis. Front Neurol. 2021; 11: 615706. DOI: 10.3389/fneur.2020.615706 EDN: YNTSAW
14. Wissel J., Bensmail D., Ferreira J. J., et al. Safety and efficacy of incobotulinum toxin A doses up to 800 U in limb spasticity: The TOWER study. Neurology. 2017; 88 (14): 1321-1328. DOI: 10.1212/WNL.0000000000003789 EDN: YXXVAD
15. Wissel J., Camões-Barbosa A., Comes G., et al. Pain Reduction in Adults with Limb Spasticity Following Treatment with IncobotulinumtoxinA: A Pooled Analysis. Toxins (Basel). 2021; 13 (12): 887. DOI: 10.3390/toxins13120887 EDN: KBSFJG
16. Костенко Е. В.,Петрова Л. В.,Ганжула П. А. и др. Опыт применения Rсеомина в коррекции спастичности руки в ранний восстановительный период инсульта. Журнал неврологии и психиатрии им. С. С. Корсакова. 2012; 112; 2: 29-34. EDN: PBZBVL
17. Коваленко А. П., Вознюк И. А., Наумов К. М. и др. Лечение спастичности у пациентов с последствиями черепно-мозговой травмы. Неврология, нейропсихиатрия, психосоматика. 2022. 14 (1): 26-31. DOI: 10.14412/2074-2711-2022-1-26-31 EDN: XSFQFT
18. Peter P. Urban, Thomas Wolf. http://ahajournals.org.
19. Хатькова С. Е., Орлова О. Р., Боцина А. Ю. и др. Основные принципы ведения пациентов с нарушением мышечного тонуса после очагового повреждения головного мозга. Consilium Medicum. 2016. 18 (2.1): 25-33. EDN: WBDETP
20. Hefter H., Rosenthal D., Jansen A., et al. Significantly lower antigenicity of incobotulinumtoxin than abo- or onabotulinumtoxin. Journal of Neurology. 2023. 270 (2): 788-796. DOI: 10.1007/s00415-022-11395-2 EDN: EFTOBE